SoloVPE Application: Polysorbate Analysis
CTech™ SoloVPE 系统采用可变光程技术,PS80 和 PS20 原液无需稀释即可进行测量,并且可提供高度可重现的结果。
Quantify Your PS80 Stock Solution
Reproducible results, without dilution
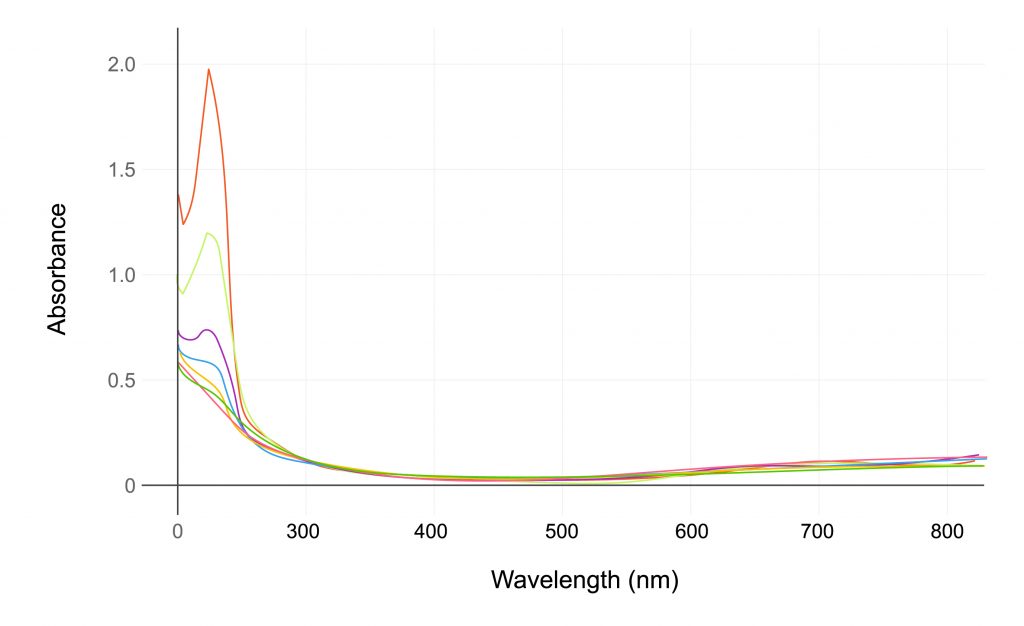
Drug product (DP) manufacturing must accurately quantify polysorbate 80 (PS80) stock solutions. However, this application can often be time-intensive and expensive. The CTech™ SoloVPE® System enables the PS80 stock solution to be measured without dilution and with reproducible results.
挑战
- Prepared in water as a stock solution, PS80 is added to the bulk drug substance (BDS). At times, this process can be unreliable — expediently quantifying the volume of surfactant present can be difficult.
- PS80’s viscous, lipophilic nature presents a challenge in accurately weighing and assessing its dissolution in water.
- Excess amounts of PS80 can be toxic at high concentrations.
- Current quantification assays can take approximately two days for results.
- Current methods require at least 30- to 50-fold dilutions of PS80 stock solution before measuring, which can result in large errors.
- Hazardous substances such as methylene chloride or cobalt may be introduced to the process.
The SoloVPE Solution
- Variable pathlength technology allows the user to measure a very wide range of polysorbate 80 and 20.
- Little to no method development is necessary depending on the concentration of the material.
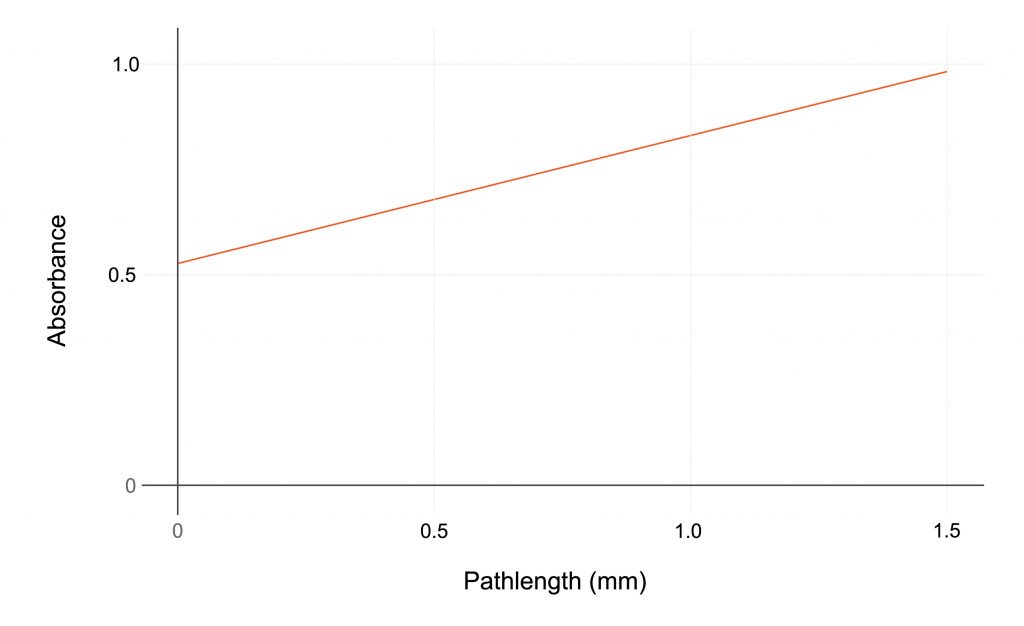
- The Slope Spectroscopy® method reduces the time from sample preparation to reproducible results from days to hours, enhancing GMP and GLP practices.
- The System provides highly reproducible results with a variable pathlength allowing an R2 of ≥0.999.
- The System is an easy inter- and intra-transfer of methods.
- The instrument is highly sensitive and able to measure from 0.0001% to 25% PS80 stock solution and 0.0001% to 100% PS20
There are no hazardous substances associated with our System so your lab and work space are safe.
Application Services
Your success is our success
CTech is committed to our customers’ success. We design service offerings to supplement our standard support options and provide you with increased access to our highly knowledgeable and experienced professionals. We offer implementation, development, and educational guidance to maximize the benefits of your variable pathlength solutions and the Slope Spectroscopy® technique.
Application Services
- 21 CFR Part 11 Guidance
- Annex 11 Guidance
- Validation Support
- Qualification Service
- Method Transfer Service
开发
- Method Design & Development
- Comparability
- On-Site Support
- Custom Standards
- Process Auditing
Education
- SOP Document Review
- Advanced Slope Spectroscopy Training
- Data Analysis
- Maintenance & Troubleshooting